Table of Contents
Introduction
The term hcooch ch2 h2o may look like a long string of letters, but it’s actually something exciting in chemistry. These letters tell us about a chemical reaction involving molecules like methyl formate and water.
This reaction is simple but powerful. It’s an example of how water can break down chemicals—a process called hydrolysis. Knowing how it works can help us learn about reactions that happen in real life, from your kitchen to your body.
In this article, we’ll take a fun, friendly approach to understanding hcooch ch2 h2o. We’ll break it down into easy steps, explore its real-world uses, and answer the most common questions you might have.
hcooch ch2 h2o and What the Formula Stands For
To understand hcooch ch2 h2o, let’s break it down:
- HCOOCH₃ is methyl formate, a fruity-smelling chemical known as an ester.
- CH₂ is part of the molecular group we study in reactions—often used to explain patterns.
- H₂O is water, the molecule we all know.
When these come together in a reaction, it means that methyl formate interacts with water and produces new chemicals. This type of change is called ester hydrolysis, meaning an ester is broken down by water. It’s simple chemistry that makes a big difference in our everyday world.
hcooch ch2 h2o Shows a Hydrolysis Reaction
In a hcooch ch2 h2o hydrolysis reaction, water helps break apart methyl formate. What’s cool is that water doesn’t just sit there—it takes action. It splits apart molecules and turns them into two smaller, useful parts.
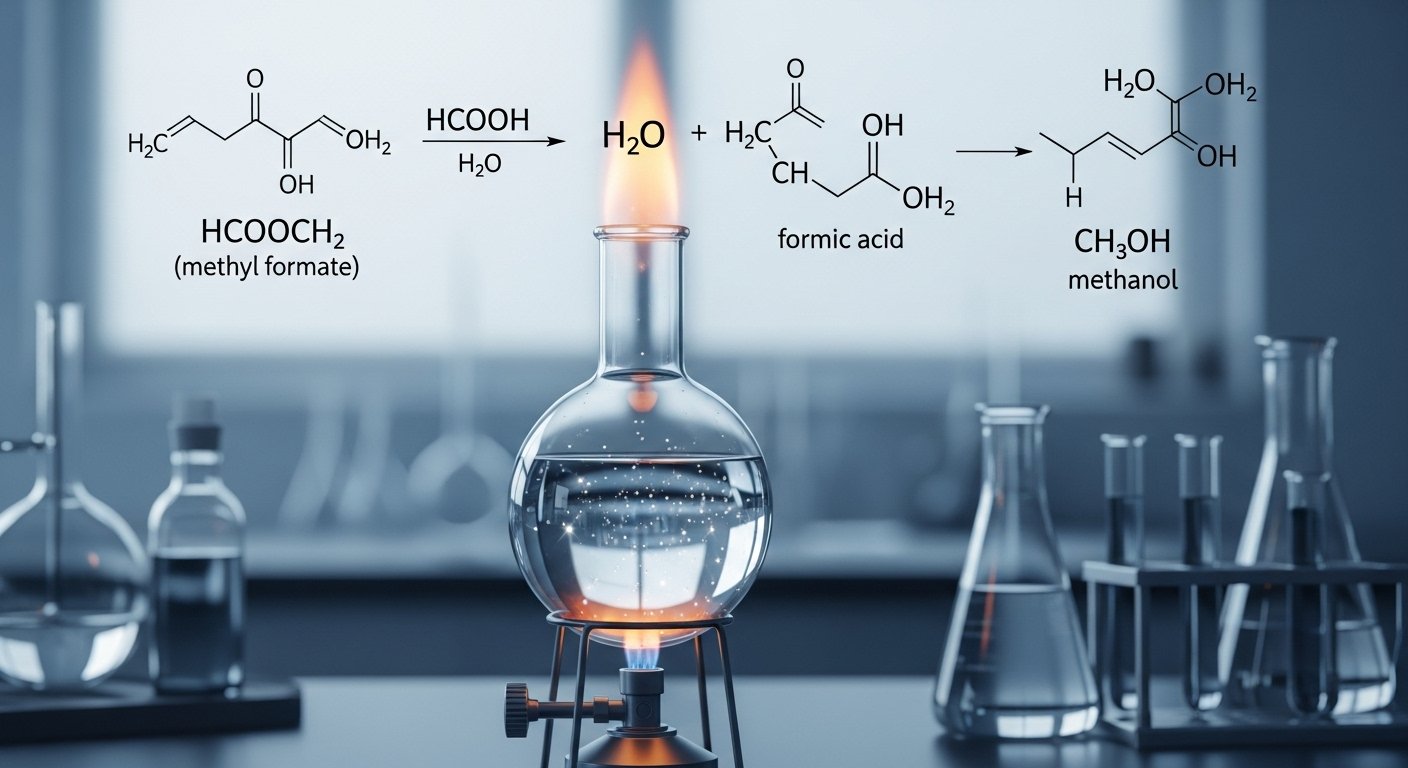
For example, methyl formate (HCOOCH₃) and water (H₂O) react to form formic acid (HCOOH) and methanol (CH₃OH). Here’s what it looks like in a simple way:
HCOOCH₃ + H₂O → HCOOH + CH₃OH
This shows how just adding water can change one thing into two useful chemicals. Hydrolysis reactions like this happen in soaps, perfumes, and even your body!
hcooch ch2 h2o and the Role of Methyl Formate
The main part of the hcooch ch2 h2o reaction is a chemical called methyl formate. It’s a type of ester, created from formic acid and methanol. It smells fruity and is found in perfumes and some flavorings.
In the reaction, methyl formate is broken down by water. Why is that important? Because it teaches us how chemical compounds can be taken apart, studied, and reused to make new things.
Understanding methyl formate is also useful in industries like plastics, paints, and fuels. So, whenever you see hcooch ch2 h2o, just remember—it’s water working its magic on a compound like methyl formate.
hcooch ch2 h2o Helps Create Formic Acid
When hcooch ch2 h2o react together, one of the new chemicals formed is formic acid. This acid might sound scary, but it’s common in nature. Ants produce it in their venom, and it’s also found in nettles.
In fact, scientists named it “formic acid” because “formica” is Latin for ant. In industry, it helps make rubber, food preservatives, leather treatments, and even cleaning products.
In this reaction, formic acid shows us how small molecules can serve important jobs. So, the hcooch ch2 h2o combination isn’t just a formula—it’s a doorway to everyday science.
hcooch ch2 h2o Reaction Also Produces Methanol
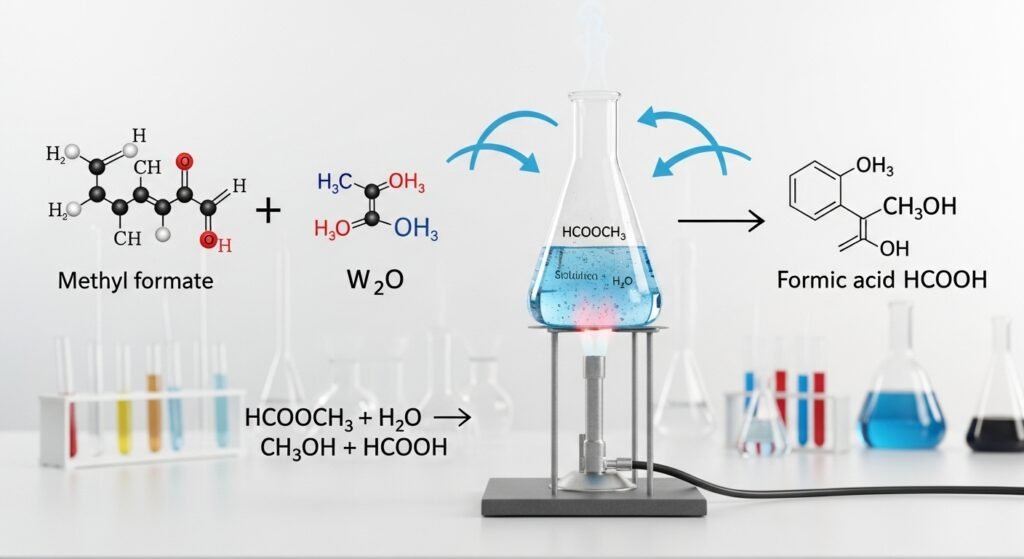
Another product formed during the hcooch ch2 h2o reaction is methanol, a kind of alcohol. Even though you can’t drink it—because it’s poisonous—methanol has many uses.
It’s used in car fuel, paints, window cleaners, and even in chemical labs. Methanol is a basic building block for creating bigger compounds. Without it, many items we use daily wouldn’t exist the same way.
So, in this reaction, water and methyl formate come together to help create methanol—a small compound with a big job in science and industry.
hcooch ch2 h2o Demonstrates the Power of Water in Chemistry
Water is everywhere, but in hcooch ch2 h2o, we see its awesome power. It’s not just something to drink or wash with. Water is a chemical that can break apart molecules.
That’s right—water helps split methyl formate into two different compounds. This process is essential to life, cooking, cleaning, and nature. It’s called hydrolysis.
So when you imagine hcooch ch2 h2o, think of water as the hero. It’s small, but it’s strong and it gets the job done—every single time.
hcooch ch2 h2o Happens in Nature and Inside Us
Did you know reactions like hcooch ch2 h2o also happen in your body? Yes! Our body uses hydrolysis to digest food, break down fats, and clean out toxins.
Nature uses hydrolysis too. Plants break down nutrients with water. Soil absorbs fertilizer using hydrolytic reactions. Even old leaves rot because of water’s effect on molecules.
That’s why learning about hcooch ch2 h2o helps us see how chemical reactions are at work around us every day—in small and powerful ways.
hcooch ch2 h2o Is Used in Lab Experiments
If you’re learning chemistry in school or working in a lab, hcooch ch2 h2o makes a great experiment. You can watch how methyl formate reacts with water to form new products.
To do this safely, your lab setup needs:
- A beaker
- Methyl formate
- Warm water
- Safety goggles
- pH strips to test acidity
Over time, you’ll see how formic acid lowers the pH and methyl formate disappears. This simple reaction helps students learn about decomposition, molecule behavior, and reaction speeds. It transforms a simple formula into a hands-on science lesson.
hcooch ch2 h2o Is a Teaching Tool for Kids and Learners
Teachers love using hcooch ch2 h2o to teach beginners about chemistry. Why? Because it’s simple, clear, and real. The reaction shows basic ideas like:
- What happens when water meets a compound
- How molecules change
- The importance of inputs and outputs
Instead of memorizing boring words, students get to watch a reaction, explore diagrams, and test results. It makes chemistry come alive—and keeps learning fun.
That’s why hcooch ch2 h2o is more than a formula. It’s a bridge to better understanding.
hcooch ch2 h2o Is Useful in Industry and Real Life
When scientists and engineers think about formulas like hcooch ch2 h2o, they think about practical uses. Ester breakdown like this is used in perfumes (to release scent), soaps (to remove grease), and even biofuels (to make clean energy).
Formic acid and methanol are useful on their own too. Farmers use formic acid to keep animal feed fresh. Meanwhile, methanol powers cars, tools, and production chains.
That means a reaction like hcooch ch2 h2o is not just lab stuff. It shows how real-life problems get solved through smart chemistry.
hcooch ch2 h2o and Sustainable Chemistry
The world is changing, and people everywhere are looking for safer, greener ways to make things. Reactions like hcooch ch2 h2o can help with that.
Because they often happen with just water and little energy, hydrolysis reactions like this are eco-friendly. No harsh chemicals, no smoke clouds—just science done cleanly and wisely.
Formulas like hcooch ch2 h2o allow chemists to build better, more sustainable materials. From plant-based fuels to biodegradable plastics, reactions like these help green science grow.
FAQs
What is hcooch ch2 h2o in chemistry?
It represents a reaction where methyl formate breaks down in water, forming formic acid and methanol.
Is hcooch ch2 h2o an acid-base reaction?
No, it’s a hydrolysis reaction where water splits an ester into an acid and an alcohol.
Why do we learn about hcooch ch2 h2o in school?
Because it’s an easy example to show how water breaks down chemicals—and it explains bigger science ideas.
Are the products of hcooch ch2 h2o dangerous?
Formic acid and methanol must be handled safely, but they are useful in small, controlled amounts.
Can I do the hcooch ch2 h2o experiment at home?
Not safely. It should be done with guidance, in a lab with proper tools and protections.
Where does hcooch ch2 h2o happen in real life?
Similar reactions happen in nature, food processing, your body, and even biofuel creation.
Conclusion
The formula hcooch ch2 h2o might seem small, but it leads to BIG ideas. It helps us understand how chemicals change, how water plays a role in reactions, and how useful compounds like formic acid and methanol are made.
This reaction connects textbooks to daily life—from science labs to our bodies to the products we use each day. It’s proof that even the most basic formulas are full of discovery.
So next time you see hcooch ch2 h2o, don’t just glance past. Smile—because now, you really know what’s happening when chemistry does its magic.